First Law of Thermodynamics Explained
📑 10 slides
👁 45 views
📅 1/24/2026
Introduction to Thermodynamics
Thermodynamics studies energy transfer in systems.

2
The 1st Law Formula
- ΔU = Q - W represents energy conservation.
- ΔU: Change in internal energy of the system.
- Q: Heat added to the system (positive if absorbed).
- W: Work done by the system (positive if done on surroundings).
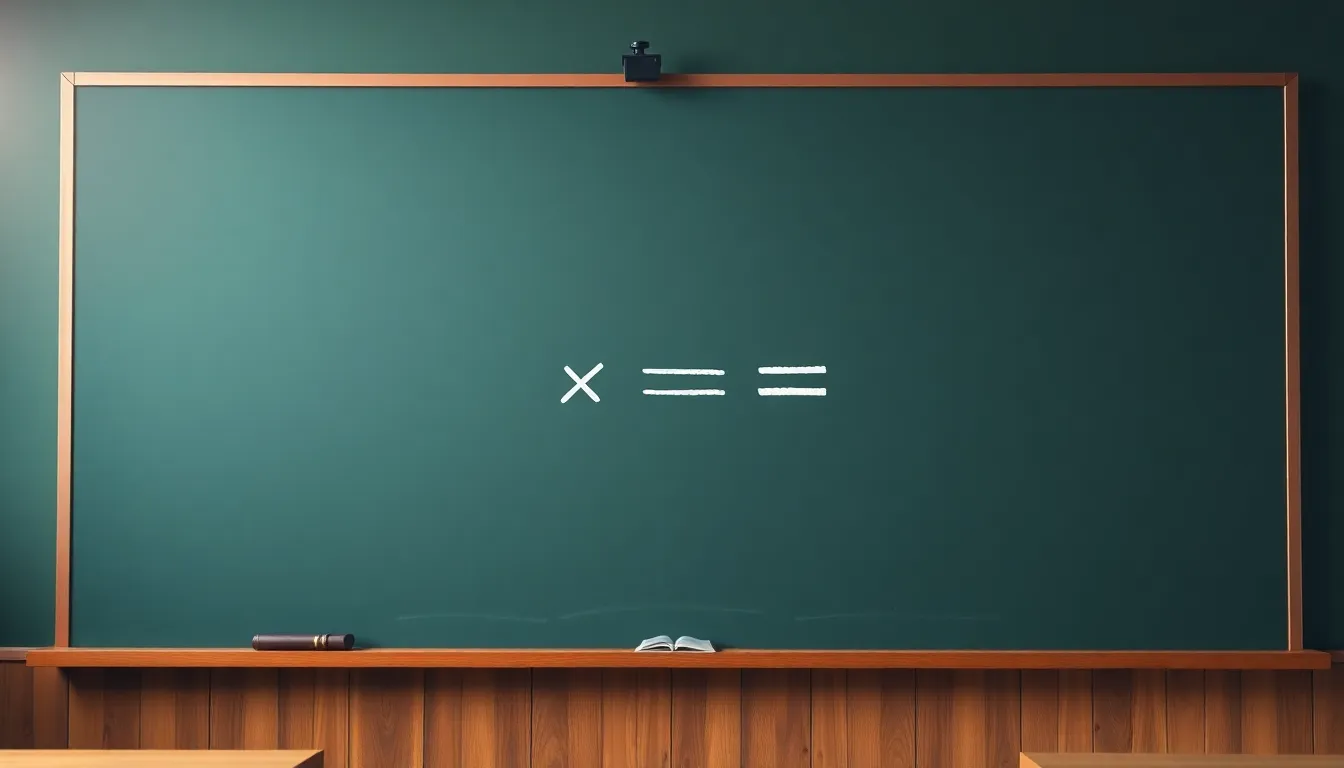
3
Understanding Internal Energy
- Internal energy (U) is the total energy of a system.
- Includes kinetic and potential energy of molecules.
- Changes with heat transfer or work done.

4
Heat Transfer (Q)
- Heat is energy transferred due to temperature difference.
- Measured in joules (J) or calories (cal).
- Positive Q: System gains heat (endothermic).

5
Work Done (W)
- Work is energy transferred by mechanical means.
- Examples: Gas expansion, piston movement.
- Positive W: System does work on surroundings.

6
Practical Applications
- Used in engines, refrigerators, and power plants.
- Explains why perpetual motion machines are impossible.
- Essential for understanding heat pumps.

7
Example: Car Engine
- Fuel combustion adds heat (Q) to the system.
- Piston movement does work (W) on surroundings.
- ΔU decreases as energy converts to motion.

8
Example: Refrigerator
- Work (W) is done to remove heat (Q) from inside.
- ΔU remains constant in a cyclic process.
- Energy is conserved but redistributed.

9
Common Misconceptions
- Energy is not lost, just transformed or transferred.
- Isolated systems have constant total energy.
- Work and heat are not properties of a system.

10
Conclusion & Summary
- 1st Law: Energy is conserved in all processes.
- ΔU = Q - W is the fundamental energy balance.
- Applies universally in physics and engineering.

1 / 10